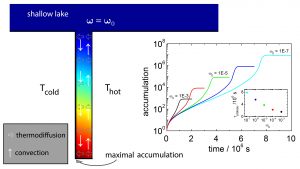
One of the central questions of humankind is: which chemical and physical conditions were necessary to make life possible? In this context, formamide plays an important role, because prebiotic molecules can be synthesized from concentrated formamide solutions. Using finite-element calculations combining thermophoresis and convection processes in hydrothermal pores, we showed that sufficiently high formamide concentrations can be accumulated to form prebiotic molecules. Depending on the initial formamide concentration, the aspect ratio of the pores and the ambient temperature, formamide concentrations of up to 85 wt % could be reached after 45–90 days, starting with an initial formamide weight fraction of 10−3 wt %, typical for concentrations in shallow lakes on early Earth. The stationary calculations show an effective accumulation only if the aspect ratio is above a certain threshold, and the corresponding transient studies display a sudden increase of the accumulation after a certain time. To explain both observations, we derived a simple heuristic model comparing the time to reach the top of the pore with the time to cross from the convective upstream towards the convective downstream. If the time to reach the top of the pore is shorter than the crossing time, the formamide molecules are flushed out of the pore. If the time is long enough, the formamide molecules can reach the downstream and accumulate at the bottom of the pore. Analyzing the optimal aspect ratio as a function of concentration, we find that at a weight fraction of 0.5, a minimal pore height is required for effective accumulation. At the same concentration, transient calculations show a maximum of the accumulation rate.
Read more: Niether D et al., PNAS.
2016;113(16):4272-4277
SoftComp partner: FZJ-Dhont